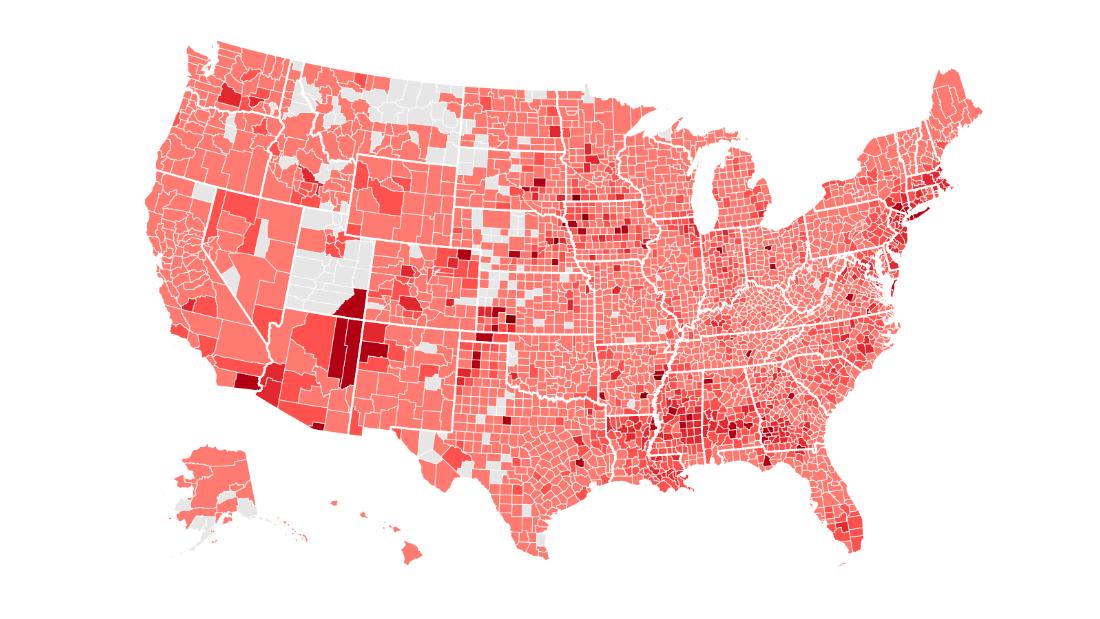
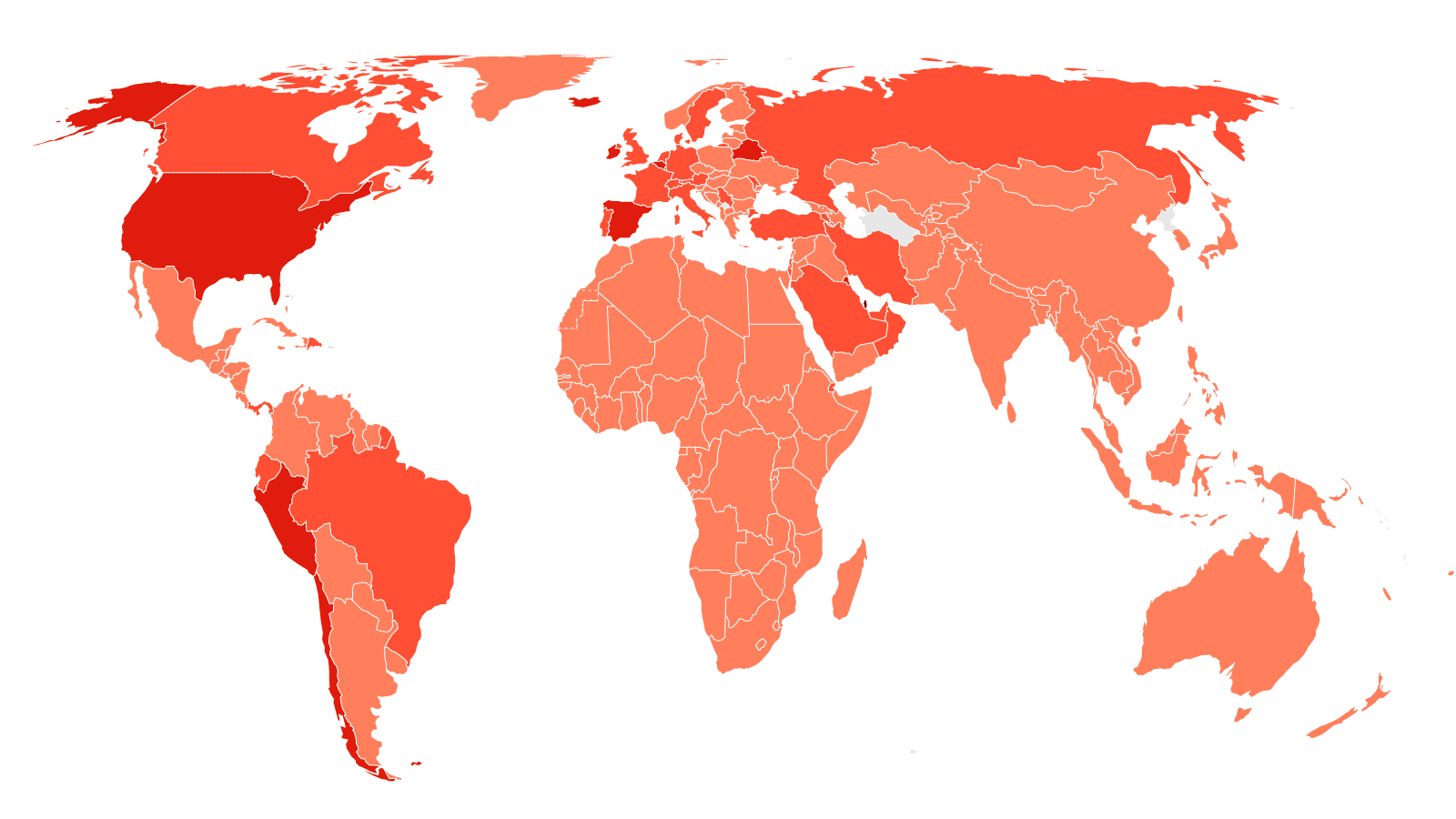
China's state-owned pharmaceutical company SinoPharm said that "hundreds of thousands" of people in the country have received Covid-19 vaccinations under an emergency use program.
The company's general counsel Zhou Song said SinoPharm has provided emergency vaccinations to high-risk groups, including medical professionals, diplomats deployed to high-risk areas, and workers for state-owned enterprises who participate in the Belt and Road Initiative, according to a statement on its website.
"(The vaccines) have been applied ... hundreds of thousands of times, and there was not a single case that demonstrated significant negative effects, nor was anyone infected," Zhou said.
In the statement, SinoPharm said it is currently developing two Covid-19 vaccines, and both vaccines are now in Phase 3 clinical trial.
The company also claimed that 100 million doses of Covid-19 vaccines can be produced by the end of this year.
Zhou said that "the vaccines were proven effective" because none of those who traveled to high-risk places after receiving the injections were infected with Covid-19.
He added that individuals receiving the vaccine will have to get two doses to generate antibodies, with the second shot coming two to four weeks after the first shot.
Wu Guizhen, chief biosecurity expert of China's CDC, told China's state-run broadcaster CGTN on Monday that Covid-19 vaccines will be ready by November.
"The public can start taking the vaccine around November or December as the performance of Phase III trials goes well," said Wu.